Pharmacotherapy
Nicotine (the drug in tobacco that causes addiction), is a powerful drug capable of inducing a variety of pharmacologic effects, including an alteration in brain chemistry, however, tobacco addiction is more than just a brain disease. It’s a complex process involving the interplay of many factors that influence an individual’s decision to use tobacco.
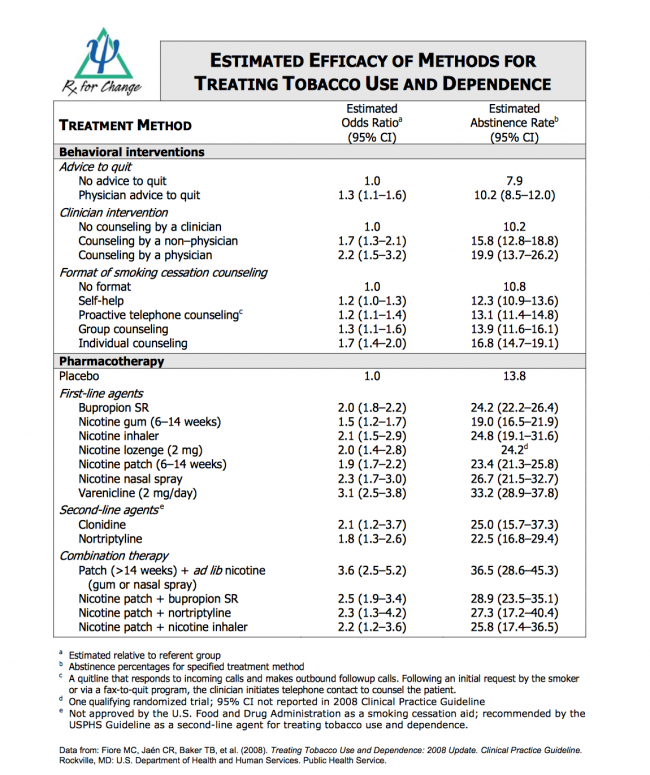
Tobacco use is clearly a complex, addictive behavior and to maximize patients’ chances of successfully quitting, clinicians should advocate behavioral interventions in combination with one or more Food and Drug Administration (FDA)-approved medications for cessation, except in cases where there may be contraindications.
Pharmacotherapies for tobacco dependence are among the most cost-effective in all of medicine and should be considered a first-line treatment. Research shows that both counseling and medicine are effective independently, but the combination is more effective than either alone.
The 2006 Morbidity and Mortality in People with Serious Mental Illness report issued by the National Association of State Mental Health Program Directors, found that persons with serious mental illness die, on average, 25 years earlier and suffer increased medical co-morbidity, often from tobacco-related disease. In fact, they are more likely to die from tobacco-related diseases than from alcohol use. These individuals may have more difficulty quitting smoking for a variety of psychological and social reasons, thus the importance to use evidence-based tobacco treatment approaches for this population.
Types of Pharmacotherapy
There are currently seven FDA-approved medications for tobacco dependence treatment. Five of which are different types of nicotine replacement therapies (NRT). The other two are non-nicotine treatments. Talk to your healthcare provider to see if these options are right for you.
Nicotine Gum (Nicorette; generics)
FDA approved: 1984
Available strengths: 2mg, 4mg
Nicotine Lozenge (Nicorette lozenge and Nicorette mini lozenge; generics)
FDA approved: 2002
Available strengths: 2mg, 4mg
Nicotine Patch (NicoDerm CQ; generics)
FDA approved: 1991
Available strengths: 7mg, 14mg, 21mg
Nicotine Nasal Spray
FDA approved: 1996 (prescription only)
Available strength: 50mcL spray with .5mg nicotine
Nicotine Inhaler (Nicotrol Inhaler)
FDA approved: 1997 (prescription only)
- Pfizer discontinued Nicotrol inhaler in the first half of 2023. The product was discontinued due to unavailability of an essential resin used to produce the inhaler mouthpiece. Additional information can be found at https://cdn.pfizer.com/pfizercom/DHCP_Letter_Nicotrol_Inhaler_5.30.2023.pdf
Bupropion SR (Zyban or Wellbutrin)
FDA approved: 1997, generic approved 2004 (prescription only)
Varenicline (Chantix)
FDA approved: 2006 (prescription only)
Benefits of Utilizing Pharmacotherapy
- Reduces physical withdrawal symptoms associated with nicotine cessation.
- Eliminates the immediate, reinforcing effect of nicotine that is rapidly absorbed via tobacco smoke.
- Allows the patient to focus on behavioral and psychological aspects of tobacco cessation.
- NRT use significantly improves the success rates of smoking cessation. Use of NRT approximately doubles long-term quit rates relative to placebo7.
- Patients are not exposed to the carcinogens and other toxic components found in tobacco and tobacco smoke.
Did you Know…
In Indiana, pharmacists can help people quit tobacco (including smoking) by providing counseling and prescribing tobacco cessation medications. This can include the nicotine patch, gum, lozenge, nasal spray, and inhaler as well as varenicline (formerly sold as Chantix) and bupropion SR (formerly sold as Zyban).
Click here to view the Standing Order issued by the Indiana State Health Commissioner which allows Indiana pharmacists to prescribe tobacco cessation products. A protocol is available which specifies the criteria and procedures to assist pharmacists in providing safe and effective tobacco cessation therapy in Indiana.